The experiment shows possibilities of gas sensor based on QCM with dissipation monitoring and AFM integrated system. The bromocresol purple (BCP) and ammonia interaction is chosen as a modeling system because BCP is well-known ammonia indicator. Hafnium dioxide nanocrystals are used for increasing the indicator surface area and for easy monitoring of surface topography changes.

Probe NanoLaboratory NTEGRA Aura performs under controlled atmosphere environments.
The sample was prepared by the following steps:
Optical microscope image of the chosen surface area Area size: 500 х 350 um
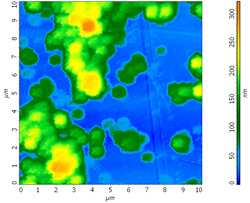
AFM topography of the chosen area Size: 10 х 10 um
Basic frequency of oscillation with the deposited substance: 5,007,536.3 Hz Quartz frequency stability on 5 minutes interval: about +/-0.06 Hz
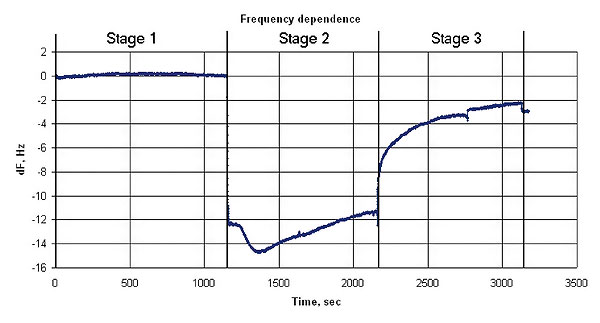
The crystal resonator with the sensor material coating is placed in a gas flow-through cell. The ammonia and nitrogen mixture is injected into the cell. The gas analyzer shows an ammonia concentration equal to 279 mg/m3 on the output of the cell. The BCP film adsorbs the ammonia that is confirmed by the resonator mass increasing (see the graphs below).
After the ammonia was stopped the system is purged with nitrogen. The desorption occurred at this stage is confirmed by the resonator coating mass decreasing and corresponding quartz resonator oscillation frequency increasing. Some of the ammonia was adsorbed irreversibly, as shown at the graphs.
The graphs below represent oscillation frequency and mass changes vs time. The mass was calculated according to the Sauerbrey model.
Quartz resonator frequency changes during the experiment
Mass changes during the experiment
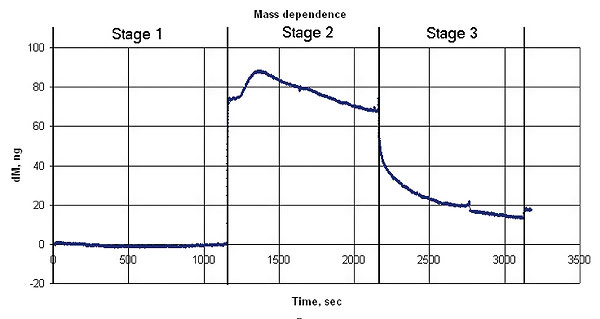
Maximum mass of the adsorbed ammonia in the experiment is 86 ng
The images below illustrate three experiment stages.
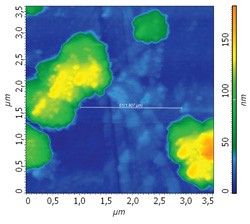
In the AFM images it is clearly seen that the topography of hafnium dioxide coated with BCP film changes under the ammonia influence.
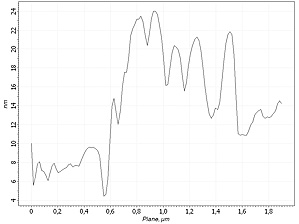
For further verification, each AFM image is coupled by cross-section profile of the same surface area at the different experiment stages.
AFM images of cluster topography: 3.5 х 3.5 um
Cluster section profile: 2 um
Stage 1. t = 0 ÷ 1200 sec
Initial condition. Ammonia absence
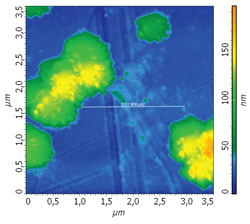
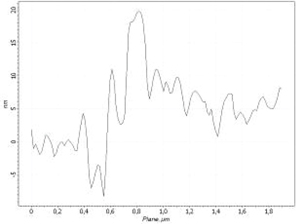
Stage 2. t = 1200 ÷ 2200 sec
Sorption during ammonia and nitrogen mixture gas injection
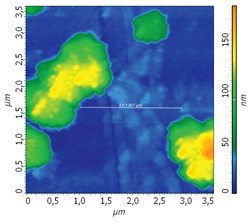
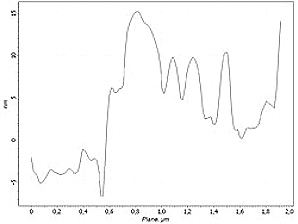
Stage 3. t = 2200 ÷ 3200 sec
Desorption while pure nitrogen injection after ammonia was stopped.
The experiment shows possibilities of the gas sensor on the base of QCM with dissipation monitoring and AFM integrated system. AFM and QCM with dissipation monitoring integration allows one to carry out simultaneous monitoring of the surface structure changes and corresponding changes of the sorbed analyte gas integral mass.
The experiment was carried out by Dr. Sevastyanov V.G., Popov V.S., Shelaev. A.V. (N.S. Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences)